Iron catalysts consistently produced the most isomerized products of the catalysts that were tested. In addition, only iron produced significant selectivities to acids, aldehydes, and ketones. All three active metals produced linear alcohols as the major oxygenated product. Carbon mole percentages of CO that are converted to specific materials for Co, Fe, and Ru catalysts vary, but they depend on support type (especially with cobalt and ruthenium) and promoters (especially with iron). Reaction tests were conducted while using an isothermal continuously stirred tank reactor. 
Discussions center on typical product selectivity of commonly used catalysts, as well as some uncommon formulations that display selectivity anomalies. The purpose of this article is to compare the product selectivity over several FTS catalysts. 
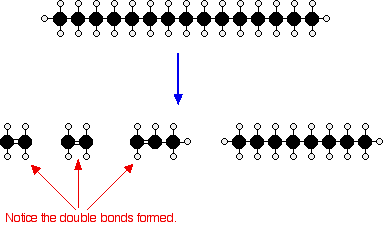
These materials can be used to probe the FTS mechanism or to produce alternative chemicals. A vast number of different oxygenated species, such as aldehydes, ketones, acids, and alcohols, are also embedded in this product range. Combined, these products provide 80–95% of the total products (excluding CO 2) generated from syngas. Minor hydrocarbon products can include isomerized hydrocarbons, predominantly methyl-branched paraffin, cyclic hydrocarbons mainly derived from high-temperature FTS and internal olefins. Additional hydrocarbon products, which can also be a majority, are linear olefins, specifically: 1-olefin, trans-2-olefin, and cis-2-olefin. The bulk of the products that were synthesized from Fischer–Tropsch synthesis (FTS) is a wide range (C 1–C 70+) of hydrocarbons, primarily straight-chained paraffins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
